Chemically enhanced nanographene molecules
16 Jul 2013
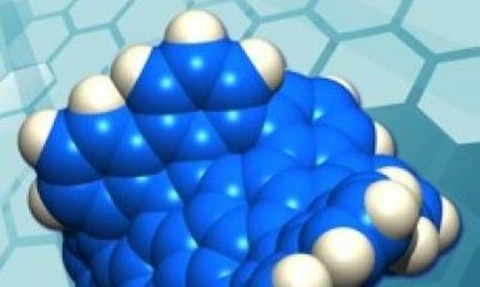
Experts from Boston College and Nagoya University have synthesised a new form of carbon that consists of grossly warped graphene.
The innovative material is constructed of multiple pieces of identical graphene that have been warped via chemical synthesis.
Each of the pieces contain 80 carbon atoms that are joined together in a network of 26 rings, with a further 30 hydrogen atoms which hug the outer surface.
Grossly warped nanographene is dramatically more soluble than a planar nanographene
The individual molecules measure slightly more than a nanometre and have been dubbed ’grossly warped nanographenes’.
Since discovering the possibility to join carbon atoms to form hollow balls, or fullerenes; scientists have been able to create ultra-thin carbon nanotubes and large, flat sheets of graphene.
The newly synthesised carbon has been distorted from usual planarity due to the presence of its five 7-membered rings and one 5-membered ring embedded in the hexagonal lattice of carbon atoms.
Lawrence T. Scott, principle author of the report released in the online journal, Nature Chemistry, stated that odd-membered-ring defects such as these not only distort the sheets of atoms away from planarity, they also alter the physical, optical, and electronic properties of the material.
“Our new grossly warped nanographene is dramatically more soluble than a planar nanographene of comparable size,” noted Scott.
The research also revealed that the planar and the warped structures have equal oxidisation, but the warped nanographene is somewhat more difficult to reduce.
Furthermore, the research team has demonstrated that the electronic properties can be modified in a predictable manner through precisely controlled chemical synthesis.
In terms of introducing the material into the public sector, Scott and his team believe the unique make-up of the graphene is best suited for use within nanoscale electronics.

