Taking imaging to new depths
24 Jan 2013
Multiphoton microscopy has enabled major breakthroughs in deep tissue imaging. Now, one UK company is planning to take the technology to the masses.
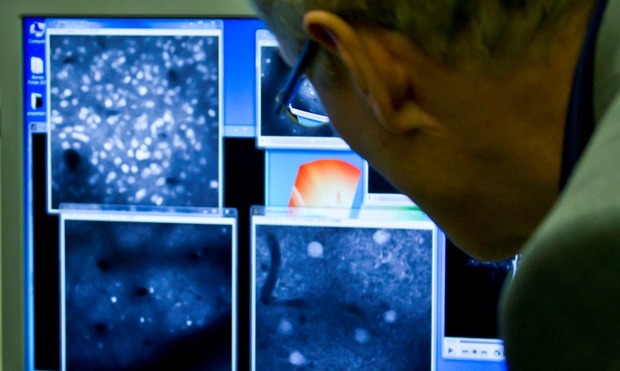
In a tiny laboratory, deep inside the endless corridors of Imperial College London, three scientists, two journalists and camera crew huddle around what is claimed to be the future of imaging.
“This is really exciting,” said Dr Simon Schultz, director of the Neural Coding Lab at Imperial, “The physics is a little bit different. It allows you to know exactly the depth from which light is emitted, which means you can image activity in the brain in real-time.”
Schultz has been working with Uckfield-based group, Scientifica, to tailor its latest multiphoton imaging system to his research. The small company, which started in 1997, last year won the Queen’s Award for Enterprise. Its success, according to managing director, Mark Johnson, is partly down to the development of the modular design of its multiphoton imaging system.
The technology has been around for some time. It was pioneered by Cornell University’s Winfried Denk in 1990 when he combined laser scanning microscopy with two-photon excitation. His multiphoton imaging system- also known as two-photon microscopy- was far more protective of the tissue being examined than confocal laser-scanning microscopy.
The multiphoton technique is very powerful and we think it is a technique that is going to become very exciting in the future
The discovery meant that for the first time, scientists could look deep into scattering tissue at depths of up to 1000 µm (1mm) for an extended period of time.
While the technology generated waves of interest among researchers, the high-cost of the equipment meant it remained the preserve of well-funded labs. This all changed in 2009 when the patent on Denk’s system expired. Scientifica was one of the companies to recognise its significance and has since developed what it claims to be a far more accessible and affordable system based on the same principles.
“The multiphoton technique is very powerful and we think it is a technique that is going to become very exciting in the future,” said Johnson. “It already has a large following throughout the world, but as the price comes down, more labs are able to use it…We’re electrophysiologists and we’re seeing imaging and physiology colliding together. If researchers want to have papers appear in top publications they need to have innovative new tools and ideas, such as combining electrophysiology with multiphoton.”
Multiphoton imaging integrates the optical process of laser scanning microscopy with long wavelength multiphoton fluorescence excitation. The technique uses high-intensity femtosecond pulsed Infrared (IR) lasers, fast-moving mirrors and precision optics to capture image structure and activity within living tissue. A laser spot is scanned quickly over a sample, exciting dyes within the tissue.
The emitted light is then collected with photomultiplier tubes (PMTs) with the resulting image being captured and displayed on a screen.
“The main difference is the light source that it uses,” explained product manager, Dale Elgar. “Normally when you use visible light, the amount of energy per photon is very high because you use a very short wavelength. But when you use infrared, the wavelength increases but similarly the power decreases. So the energy you have per photon is not enough to excite the dye.”
Elgar added that the light source is pulsed at 80 MHz, causing the energy to be focused on a single plane. “Compare this to confocal microscopy which provides good resolution but not at great depths,” said Elgar.
“That doesn’t bode well for people who want to image in the brain because the brain is a thick dense piece of tissue. That’s where multiphoton comes into its own because infrared can penetrate the tissue a lot better. It can go down 500-700 microns. That is the main difference and also probably the limiting step in multiphoton because the price of these lasers is very expensive.”
Elgar and his team have brought the cost down by taking a modular approach to its design. The central components in Scientifica’s system are the Multiphoton Detection Unit (MDU) and scanhead, which achieves diffraction limited resolution.
We’re seeing imaging and physiology colliding together
The modular approach means that the system allows integration with other experimental techniques such as electrophysiology. The group has also chosen to use a range of free open-source software packages that can be developed for a particular application by the user.
“Our development has come in stages,” said Elgar. “We first developed an upright microscope which was used for purely electrophysiology. Then we developed modules which we have provided as add-ons to this microscope so that users can upgrade to multiphoton. It’s a modular concept, so you can buy parts and you can build the other parts yourself. Maintaining this modular approach is high on our list of priorities for the future.”
Back at Imperial, Schultz is keen for the technology to become available to a wider range of research scientists. His own lab has been working on reverse engineering the information processing architecture of the brain. Multiphoton imaging, along with optogenetic manipulation and electrophysiology allow to measure and interfere with patterns in the neuronal activity in mice. But the range of potential applications is huge.
“We’re still learning just how multiphoton can help researchers in their work,” said Johnson. “New applications are appearing all the time, and we’re very excited about the future.”