Head towards the light
14 May 2015
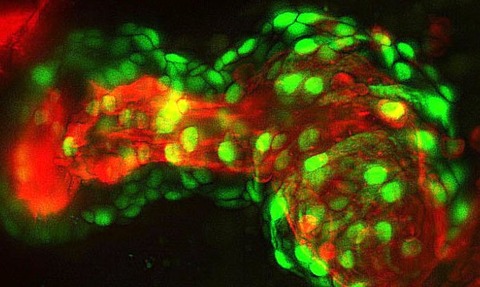
Advances in light sheet microscopy are opening an entirely new world of discovery for the imaging research community.
Light sheet fluorescence microscopy (LSFM) - often referred to as selective plane illumination microscopy - is an imaging technique used to side-illuminate a fluorescently tagged biological sample with a thin section of relatively low-power excitation light.
The concept of LSFM was originally developed in 2004 in Professor Ernst Stelzer’s laboratory at the European Molecular Biology Laboratory in Heidelberg, Germany.
“It remains to be seen what new techniques will be added to extend the LSFM application space
Hamamatsu Photonics sales manager Jim Owens
Marco Marcello, who worked in Stelzer’s lab in 2005 and who is now light microscopy facility manager at the University of Liverpool’s Institute of Integrative Biology, says LSFM comprises a complete paradigm shift in the way microscopy specialists conduct live imaging in 3D.
“Thanks to its strategy of illumination, photodamage to all the structures that are not on the focal plane is minimised and signal-to-noise ratio is much better than in standard confocal and wide-field microscopy,” Marcello says.
“The fact that the sample being imaged is not squashed between a slide and a coverslip, but embedded into a hydrogel matrix and free to rotate, allows for a multi-view collection of the images - essentially a tomographic approach in microscopy.”
For Marcello, LSFM truly excels when applied to the imaging of living samples.
“LSFM can, for example, record the different firing patterns of single neurons in the brain of a zebrafish (immobilised in a hydrogel),” Marcello says.
And as the zebrafish being imaged is not usually killed during the initial study, it can also be observed at a later development stage in its lifecycle, he adds.
“I am not aware of another technology that is able to do this at such a high level of resolution.”
Arguably, superresolution microscopy is the only other imaging technology to possess anything like LSFM’s unique qualities.
Last year, developments in superresolution microscopy led to Eric Betzig, W.E. Moerner and Stefan Hell winning the Nobel Prize in Chemistry.
“Superresolution strives to reach a high spatial resolution of a sample, normally beyond the defraction limit of light according to Abbe, which is approximately 250 nanometres,” Marcello says.
“This high resolution is normally achieved at the expense of other factors, such as longer acquisition times and lower depth of penetration in the sample.”
However, Marcello says LSFM has an “obvious edge” over superresolution - especially when taking into account its capabilities in imaging live systems.
Unfortunately, though, LSFM suffers from a number of drawbacks.
“For example, a single time-lapse LSFM experiment watching for the development of an organoid or a fish can easily generate more than two terabytes of data,” Marcello says.
“Not all facilities are able to cope with these big data,” he says.
Jim Owens, sales manager at opto-electronic firm Hamamatsu Photonics, agrees with Marcello - suggesting the mountain of data generated by LSFM is a pressing issue that most biologists struggle with.
“Yet, being able to compress the raw data on the fly - starting at the point of image acquisition with the latest smart scientific CMOS cameras that can deliver just the areas of interest and discard the rest - to powerful offline processing that uses specialised algorithms to produce optimised datasets further downstream are some of the ways this is being addressed,” Owens says.
For Owens, we are still only at the beginning of widespread LSFM implementation.
“It remains to be seen what new techniques will be added to extend the LSFM application space and, more importantly, what new research discoveries this will lead to,” he says.
No Nobel?
Unfortunately, though Owens says advances in LSFM will likely lead to new discoveries, Marcello is unsure whether Stelzer’s contribution to microscopy will earn him a Nobel Prize.
“I think one of the factors decreasing the likelihood of a Nobel Prize for Ernst [Stelzer] is the fact that the illumination strategy of the light sheet had already been invented by Richard Adolf Zsigmondy and Henry Siedentopf with the ’ultramicroscope’ at the beginning of the 20th century,” Marcello says.
“However, the light sheet is more important than its illumination strategy,” he says.
“Many other important concepts behind the light sheet, including ideas of using that illumination strategy in connection with fluorescence microscopy, using the tomography approach and applying to the life science, are genuinely from Stelzer.”

